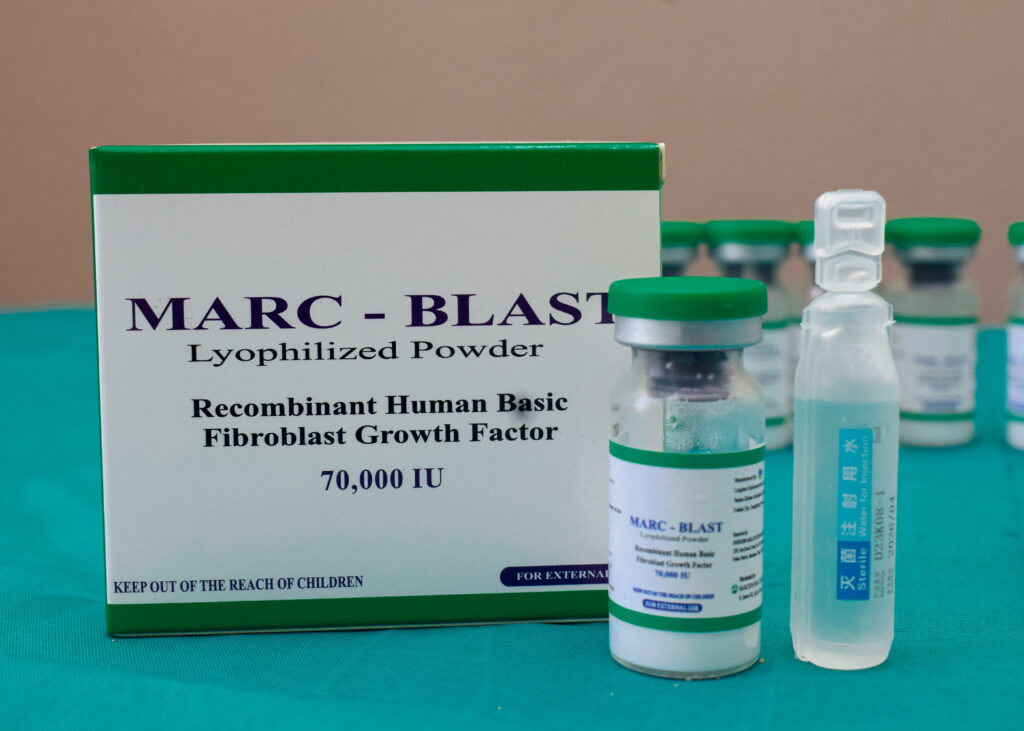
Marcblast
Composition
Each vial contains 70,000 IU of rh-bFGF
Product Description
Dossage
1. Open the aluminium cap of the vial (lyophilized powder) along the direction of arrow; unscrew the flat upper part of the plastic ampoule bottle (sterile water for injection) until they are separated.
2.Take down the rubber stopper of the vial: fetch the medicinal spray pump, tear the protective film and place upside down
3. Pour 5 ml of sterile water for injection into the vial for complete reconstitution
4. Assemble the medicinal spray pump on the vial; slightly shake well.
5. Precaution: after reconstitution, it should be stored at 2°C – 8°C and used within 7 days.
Nafdac Reg
A6–100130
Indications
Promotion of wound healing and can be used for chronic wounds (including chronic granulation wounds, ulcer and bedsore, etc.) fresh wounds (including trauma, surgical wounds,etc.) and burn wounds.
Dosage
Dosage of rh-bFGF is 150 IU/cm
Therapeutic class
Contracdiction
Hypersensitivity to rh-bFGF, or to any of the excipients, or hypersensitivity to substances with a similar action, allergic to the product should be forbidden to use the product
Presentation
1 vial of rh-bFGF Lyophilized powder
1 bottle of sterile water for injection
1 medical spray pump
Side Effects
No obvious adverse reaction is found
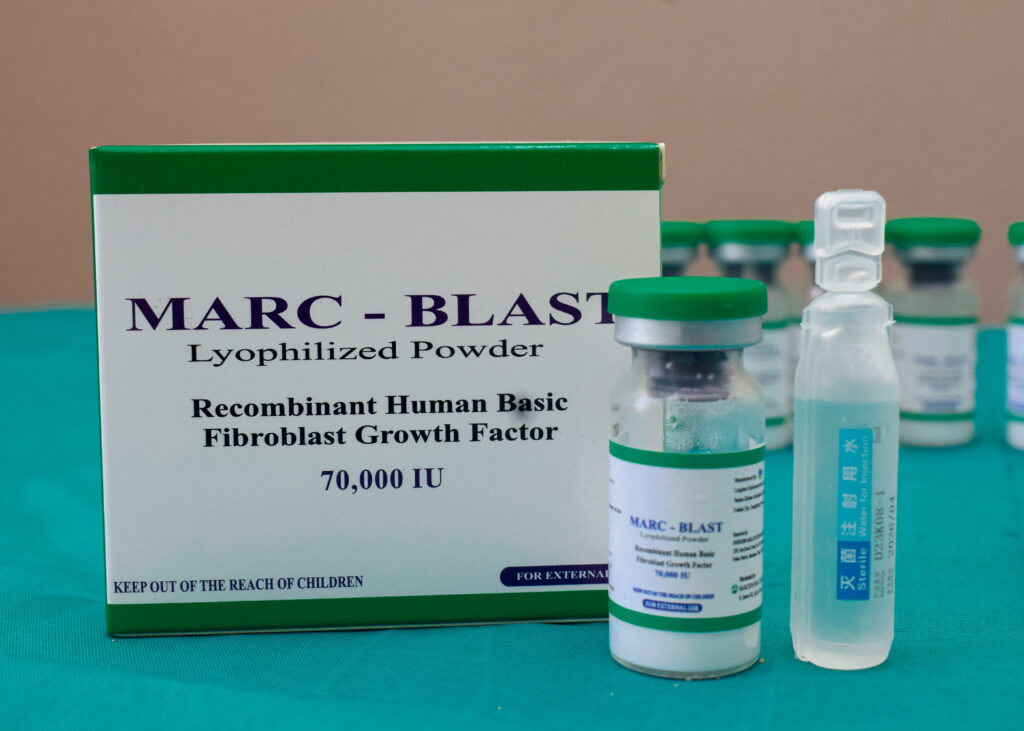
MARCAT TABLETS
Product Description
Direction for use
1. Open the aluminium cap of the vial (lyophilized powder) along the direction of arrow; unscrew the flat upper part of the plastic ampoule bottle (sterile water for injection) until they are separated.
2.Take down the rubber stopper of the vial: fetch the medicinal spray pump, tear the protective film and place upside down
3. Pour 5 ml of sterile water for injection into the vial for complete reconstitution
4. Assemble the medicinal spray pump on the vial; slightly shake well.
5. Precaution: after reconstitution, it should be stored at 2°C – 8°C and used within 7 days.
Indications
Promotion of wound healing and can be used for chronic wounds (including chronic granulation wounds, ulcer and bedsore, etc.) fresh wounds (including trauma, surgical wounds,etc.) and burn wounds.
Composition
Each vial contains 70,000 IU of rh-bFGF
Nafdac Reg
A6–100130
Dosage
Dosage of rh-bFGF is 150 IU/cm
Therapeutic class
Contracdiction
Hypersensitivity to rh-bFGF, or to any of the excipients, or hypersensitivity to substances with a similar action, allergic to the product should be forbidden to use the product
Presentation
1 vial of rh-bFGF Lyophilized powder
1 bottle of sterile water for injection
1 medical spray pump
Side Effects
No obvious adverse reaction is found
